Decoding the Immune "Signal Messenger" IFN-γ: A Comprehensive Perspective from Molecular Mechanisms to Clinical Applications
In the complex immune defense network of the human body, there is a cytokine hailed as the "key transmitter of immune signals." It can both awaken dormant immune cells and precisely target pathogens and abnormal cells—this is Interferon-γ (IFN-γ). From basic immunological research to clinical diagnosis and treatment of diseases such as tumors and infectious diseases, every molecular mechanism of IFN-γ holds the potential to break through medical bottlenecks, and every upgrade in detection technology provides a more accurate basis for disease diagnosis.
I. IFN-γ: The "Multifunctional Signal Hub" in the Immune Network
As the core member of the type II interferon family, IFN-γ was first discovered and named by scientists in 1973. Unlike type I interferons (such as IFN-α/β), which are mainly involved in innate antiviral immunity, IFN-γ is a typical "adaptive immune regulatory factor," primarily secreted by activated T lymphocytes (such as Th1 cells, cytotoxic T cells) and natural killer (NK) cells. Its molecular structure is a homodimer, which exerts effects by binding to specific receptors (IFN-γR) on the surface of target cells. This highly specific binding mode ensures the precision of immune signal transmission and avoids immune response disorders.
As a "central node" of the immune network, IFN-γ is unique in that it can connect innate immunity and adaptive immunity and regulate the differentiation and function of immune cells. This "connecting link" characteristic makes it a "star molecule" in the field of immunological research.
II. Signaling Pathway: A Precisely Regulated "Molecular Conduction Chain"
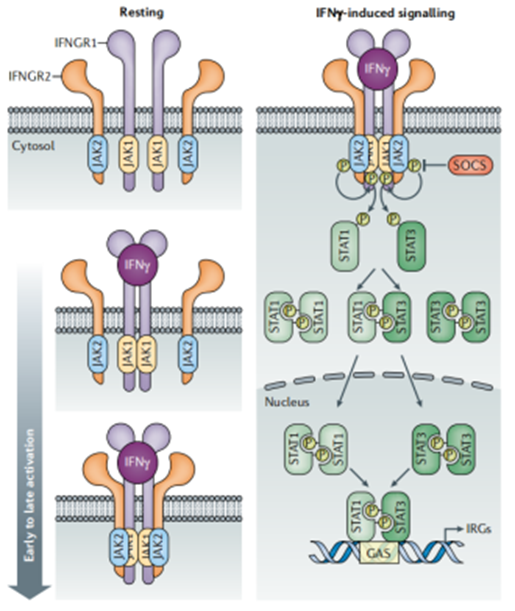
The biological functions of IFN-γ rely on a highly conserved and sophisticated signal transduction pathway—the JAK-STAT pathway. Its transduction process is like a "precise and efficient molecular circuit," with each step strictly regulated to ensure signals are not interfered with or mistransmitted.
When the IFN-γ homodimer binds to the IFN-γR1/IFN-γR2 heterodimeric receptor on the target cell membrane, it first activates the JAK kinases (JAK1 and JAK2) coupled to the intracellular domain of the receptor. The activated JAK kinases undergo autophosphorylation and further phosphorylate the tyrosine residues of the receptor, forming "signal anchor sites." Subsequently, STAT1 molecules in the cytoplasm are recruited to the phosphorylated sites of the receptor and undergo tyrosine phosphorylation catalyzed by JAK kinases. The phosphorylated STAT1 molecules rapidly form homodimers (i.e., GAF complexes).
The formed GAF complex possesses nuclear localization ability, enabling it to cross the nuclear membrane and enter the nucleus, where it specifically binds to the "Interferon-γ activation sequence (GAS)" in the promoter region of target genes. This initiates the transcriptional expression of a series of downstream genes, including cytokine genes that regulate immune cell activation, antiviral genes that inhibit pathogen replication, and tumor suppressor genes involved in apoptosis regulation. Notably, there are also negative regulators in this pathway (such as SOCS family proteins) that can timely terminate signal transduction, preventing excessive activation of immune responses from damaging normal cells, which reflects the balance of biological regulation.
III. Core Functions: A "Versatile Player" in Immune Defense and Cell Regulation
The functions of IFN-γ cover multiple dimensions such as immune defense and cell homeostasis regulation, and the diversity of its mechanisms of action endows it with broad application potential in the field of biomedicine.
In terms of immune defense, IFN-γ is a "core weapon" against intracellular pathogens (such as Mycobacterium tuberculosis, hepatitis B virus, HIV, etc.). It can activate the phagocytic and killing functions of macrophages, promote macrophages to produce reactive oxygen species (ROS) and nitric oxide (NO), and enhance the ability to clear intracellular bacteria and viruses. At the same time, IFN-γ can promote the maturation of dendritic cells, enhance their antigen-presenting ability, thereby activating more specific T lymphocytes to participate in immune responses and forming "sustained immune memory."
In tumor immune regulation, IFN-γ exerts a "dual role": on the one hand, it can directly inhibit the proliferation of tumor cells, induce tumor cell apoptosis, and at the same time upregulate the expression of MHC class I molecules on the surface of tumor cells, enhance the "immunogenicity" of tumor cells, making them more easily recognized and killed by cytotoxic T cells; on the other hand, IFN-γ can regulate the tumor microenvironment, inhibit the activity of immunosuppressive cells (such as Treg cells, M2-type macrophages), break the "immune escape" barrier of tumors, and provide a "favorable environment" for tumor immunotherapy.
In addition, IFN-γ also plays an important role in immune tolerance regulation and inflammatory response balance. It can regulate the differentiation balance of Th1/Th2 cell subsets, avoiding allergic diseases caused by excessive activation of Th2 cells; at the same time, abnormal expression of IFN-γ is closely related to the occurrence and development of autoimmune diseases, reflecting its "double-edged sword" characteristic.
IV. Disease Association: A "Key Biomarker" for Clinical Diagnosis and Treatment
The expression level of IFN-γ is closely related to the occurrence, development, severity, and prognosis of various diseases, making it a highly valuable "biomarker" and therapeutic target in clinical diagnosis and treatment.
In the field of infectious diseases, IFN-γ detection is one of the "gold standards" for the diagnosis of Mycobacterium tuberculosis infection. After Mycobacterium tuberculosis infection, specific T cells in the body secrete IFN-γ under antigen stimulation. By detecting the level of IFN-γ secreted by specific T cells in peripheral blood (such as T-SPOT.TB assay), latent tuberculosis infection can be diagnosed quickly and accurately, avoiding the defects of traditional sputum culture methods such as long time consumption and low sensitivity. In viral hepatitis (such as hepatitis B and C), the expression level of IFN-γ is positively correlated with the degree of liver inflammation. High expression of IFN-γ indicates active antiviral immune response in the body, which can provide a reference for the selection of clinical treatment plans.
In the field of oncology, the expression pattern of IFN-γ is closely related to tumor prognosis. Studies have shown that patients with higher IFN-γ levels in the tumor microenvironment have better curative effect when receiving immune checkpoint inhibitor (such as PD-1/PD-L1 inhibitor) treatment, because IFN-γ can enhance the immune activity of the tumor microenvironment and improve the response rate of immunotherapy. In addition, abnormal activation or inhibition of the IFN-γ signaling pathway is also closely related to tumorigenesis. For example, STAT1 gene mutations leading to defects in the IFN-γ signaling pathway will increase the risk of tumor occurrence.
In autoimmune diseases, abnormally high expression of IFN-γ is one of the important pathogenic factors. In diseases such as rheumatoid arthritis and systemic lupus erythematosus, immune cells secrete excessive IFN-γ, leading to aggravated inflammatory responses and damage to tissues and organs. Therefore, drugs targeting the IFN-γ signaling pathway (such as anti-IFN-γ monoclonal antibodies) have become a new direction for the treatment of autoimmune diseases.
V. Detection Technologies: "Precision Tools" from Basic Research to Clinical Applications
With the development of biotechnology, IFN-γ detection technologies have been continuously upgraded. From early bioactivity assays to current molecular-level detection, the sensitivity, specificity, and timeliness of detection have been significantly improved, providing strong support for basic research and clinical diagnosis and treatment.
Traditional detection methods are mainly bioactivity assays, which indirectly quantify IFN-γ levels by detecting the antiviral activity or proliferation inhibitory effect of IFN-γ on sensitive cell lines (such as L929 cells). This method can reflect the biological function of IFN-γ, but it is complex to operate, time-consuming, and easily interfered by other cytokines, and has gradually been replaced by more precise methods.
Enzyme-linked immunosorbent assay (ELISA) is currently the most widely used IFN-γ detection method in clinical practice. Based on the principle of specific antigen-antibody binding, it realizes quantitative detection of IFN-γ through enzyme-labeled antibodies. This method is simple to operate and highly specific, and can quickly detect IFN-γ levels in serum, plasma, and other samples, which is widely used in the auxiliary diagnosis and curative effect monitoring of infectious diseases.
For the detection of IFN-γ at the cellular level, enzyme-linked immunospot assay (ELISPOT) and flow cytometry (FCM) have become mainstream technologies. ELISPOT can detect the ability of a single cell to secrete IFN-γ with extremely high sensitivity, and can be used for specific diagnosis of tuberculosis infection, screening of tumor-specific T cells, etc.; flow cytometry detects intracellular IFN-γ expression through fluorescently labeled antibodies, and can simultaneously analyze the secretion of IFN-γ by different cell subsets (such as Th1 cells, NK cells), providing more detailed information for immune function evaluation.
At the molecular level, quantitative real-time PCR (qPCR) technology can reflect the transcriptional activity of IFN-γ by detecting the mRNA expression level of the IFN-γ gene, which is used in early disease diagnosis, pathogenesis research, and other fields. In addition, the development of new detection technologies based on microfluidic chips and electrochemical sensors has further realized rapid and micro-detection of IFN-γ, making point-of-care testing (POCT) possible.
Conclusion: IFN-γ—the "Bridge Molecule" Connecting Basic Research and Clinical Applications
From the precise transmission of immune signals to the clinical diagnosis and treatment of diseases, every molecular characteristic of IFN-γ demonstrates the charm of biomedicine. With the in-depth analysis of its signaling pathway mechanisms, continuous innovation of detection technologies, and breakthroughs in the development of targeted drugs, the application prospect of IFN-γ in infectious diseases, tumors, autoimmune diseases, and other fields will be broader. In the future, with the deep integration of multi-omics technologies, gene editing technologies, and IFN-γ research, we will further uncover the mystery of its molecular mechanisms, allowing this immune "signal messenger" to play a greater role in safeguarding human health.
I. IFN-γ: The "Multifunctional Signal Hub" in the Immune Network

As the core member of the type II interferon family, IFN-γ was first discovered and named by scientists in 1973. Unlike type I interferons (such as IFN-α/β), which are mainly involved in innate antiviral immunity, IFN-γ is a typical "adaptive immune regulatory factor," primarily secreted by activated T lymphocytes (such as Th1 cells, cytotoxic T cells) and natural killer (NK) cells. Its molecular structure is a homodimer, which exerts effects by binding to specific receptors (IFN-γR) on the surface of target cells. This highly specific binding mode ensures the precision of immune signal transmission and avoids immune response disorders.
As a "central node" of the immune network, IFN-γ is unique in that it can connect innate immunity and adaptive immunity and regulate the differentiation and function of immune cells. This "connecting link" characteristic makes it a "star molecule" in the field of immunological research.
II. Signaling Pathway: A Precisely Regulated "Molecular Conduction Chain"

The biological functions of IFN-γ rely on a highly conserved and sophisticated signal transduction pathway—the JAK-STAT pathway. Its transduction process is like a "precise and efficient molecular circuit," with each step strictly regulated to ensure signals are not interfered with or mistransmitted.
When the IFN-γ homodimer binds to the IFN-γR1/IFN-γR2 heterodimeric receptor on the target cell membrane, it first activates the JAK kinases (JAK1 and JAK2) coupled to the intracellular domain of the receptor. The activated JAK kinases undergo autophosphorylation and further phosphorylate the tyrosine residues of the receptor, forming "signal anchor sites." Subsequently, STAT1 molecules in the cytoplasm are recruited to the phosphorylated sites of the receptor and undergo tyrosine phosphorylation catalyzed by JAK kinases. The phosphorylated STAT1 molecules rapidly form homodimers (i.e., GAF complexes).
The formed GAF complex possesses nuclear localization ability, enabling it to cross the nuclear membrane and enter the nucleus, where it specifically binds to the "Interferon-γ activation sequence (GAS)" in the promoter region of target genes. This initiates the transcriptional expression of a series of downstream genes, including cytokine genes that regulate immune cell activation, antiviral genes that inhibit pathogen replication, and tumor suppressor genes involved in apoptosis regulation. Notably, there are also negative regulators in this pathway (such as SOCS family proteins) that can timely terminate signal transduction, preventing excessive activation of immune responses from damaging normal cells, which reflects the balance of biological regulation.
III. Core Functions: A "Versatile Player" in Immune Defense and Cell Regulation
The functions of IFN-γ cover multiple dimensions such as immune defense and cell homeostasis regulation, and the diversity of its mechanisms of action endows it with broad application potential in the field of biomedicine.
In terms of immune defense, IFN-γ is a "core weapon" against intracellular pathogens (such as Mycobacterium tuberculosis, hepatitis B virus, HIV, etc.). It can activate the phagocytic and killing functions of macrophages, promote macrophages to produce reactive oxygen species (ROS) and nitric oxide (NO), and enhance the ability to clear intracellular bacteria and viruses. At the same time, IFN-γ can promote the maturation of dendritic cells, enhance their antigen-presenting ability, thereby activating more specific T lymphocytes to participate in immune responses and forming "sustained immune memory."
In tumor immune regulation, IFN-γ exerts a "dual role": on the one hand, it can directly inhibit the proliferation of tumor cells, induce tumor cell apoptosis, and at the same time upregulate the expression of MHC class I molecules on the surface of tumor cells, enhance the "immunogenicity" of tumor cells, making them more easily recognized and killed by cytotoxic T cells; on the other hand, IFN-γ can regulate the tumor microenvironment, inhibit the activity of immunosuppressive cells (such as Treg cells, M2-type macrophages), break the "immune escape" barrier of tumors, and provide a "favorable environment" for tumor immunotherapy.
In addition, IFN-γ also plays an important role in immune tolerance regulation and inflammatory response balance. It can regulate the differentiation balance of Th1/Th2 cell subsets, avoiding allergic diseases caused by excessive activation of Th2 cells; at the same time, abnormal expression of IFN-γ is closely related to the occurrence and development of autoimmune diseases, reflecting its "double-edged sword" characteristic.
IV. Disease Association: A "Key Biomarker" for Clinical Diagnosis and Treatment
The expression level of IFN-γ is closely related to the occurrence, development, severity, and prognosis of various diseases, making it a highly valuable "biomarker" and therapeutic target in clinical diagnosis and treatment.
In the field of infectious diseases, IFN-γ detection is one of the "gold standards" for the diagnosis of Mycobacterium tuberculosis infection. After Mycobacterium tuberculosis infection, specific T cells in the body secrete IFN-γ under antigen stimulation. By detecting the level of IFN-γ secreted by specific T cells in peripheral blood (such as T-SPOT.TB assay), latent tuberculosis infection can be diagnosed quickly and accurately, avoiding the defects of traditional sputum culture methods such as long time consumption and low sensitivity. In viral hepatitis (such as hepatitis B and C), the expression level of IFN-γ is positively correlated with the degree of liver inflammation. High expression of IFN-γ indicates active antiviral immune response in the body, which can provide a reference for the selection of clinical treatment plans.
In the field of oncology, the expression pattern of IFN-γ is closely related to tumor prognosis. Studies have shown that patients with higher IFN-γ levels in the tumor microenvironment have better curative effect when receiving immune checkpoint inhibitor (such as PD-1/PD-L1 inhibitor) treatment, because IFN-γ can enhance the immune activity of the tumor microenvironment and improve the response rate of immunotherapy. In addition, abnormal activation or inhibition of the IFN-γ signaling pathway is also closely related to tumorigenesis. For example, STAT1 gene mutations leading to defects in the IFN-γ signaling pathway will increase the risk of tumor occurrence.
In autoimmune diseases, abnormally high expression of IFN-γ is one of the important pathogenic factors. In diseases such as rheumatoid arthritis and systemic lupus erythematosus, immune cells secrete excessive IFN-γ, leading to aggravated inflammatory responses and damage to tissues and organs. Therefore, drugs targeting the IFN-γ signaling pathway (such as anti-IFN-γ monoclonal antibodies) have become a new direction for the treatment of autoimmune diseases.
V. Detection Technologies: "Precision Tools" from Basic Research to Clinical Applications
With the development of biotechnology, IFN-γ detection technologies have been continuously upgraded. From early bioactivity assays to current molecular-level detection, the sensitivity, specificity, and timeliness of detection have been significantly improved, providing strong support for basic research and clinical diagnosis and treatment.
Traditional detection methods are mainly bioactivity assays, which indirectly quantify IFN-γ levels by detecting the antiviral activity or proliferation inhibitory effect of IFN-γ on sensitive cell lines (such as L929 cells). This method can reflect the biological function of IFN-γ, but it is complex to operate, time-consuming, and easily interfered by other cytokines, and has gradually been replaced by more precise methods.
Enzyme-linked immunosorbent assay (ELISA) is currently the most widely used IFN-γ detection method in clinical practice. Based on the principle of specific antigen-antibody binding, it realizes quantitative detection of IFN-γ through enzyme-labeled antibodies. This method is simple to operate and highly specific, and can quickly detect IFN-γ levels in serum, plasma, and other samples, which is widely used in the auxiliary diagnosis and curative effect monitoring of infectious diseases.
For the detection of IFN-γ at the cellular level, enzyme-linked immunospot assay (ELISPOT) and flow cytometry (FCM) have become mainstream technologies. ELISPOT can detect the ability of a single cell to secrete IFN-γ with extremely high sensitivity, and can be used for specific diagnosis of tuberculosis infection, screening of tumor-specific T cells, etc.; flow cytometry detects intracellular IFN-γ expression through fluorescently labeled antibodies, and can simultaneously analyze the secretion of IFN-γ by different cell subsets (such as Th1 cells, NK cells), providing more detailed information for immune function evaluation.
At the molecular level, quantitative real-time PCR (qPCR) technology can reflect the transcriptional activity of IFN-γ by detecting the mRNA expression level of the IFN-γ gene, which is used in early disease diagnosis, pathogenesis research, and other fields. In addition, the development of new detection technologies based on microfluidic chips and electrochemical sensors has further realized rapid and micro-detection of IFN-γ, making point-of-care testing (POCT) possible.
Conclusion: IFN-γ—the "Bridge Molecule" Connecting Basic Research and Clinical Applications
From the precise transmission of immune signals to the clinical diagnosis and treatment of diseases, every molecular characteristic of IFN-γ demonstrates the charm of biomedicine. With the in-depth analysis of its signaling pathway mechanisms, continuous innovation of detection technologies, and breakthroughs in the development of targeted drugs, the application prospect of IFN-γ in infectious diseases, tumors, autoimmune diseases, and other fields will be broader. In the future, with the deep integration of multi-omics technologies, gene editing technologies, and IFN-γ research, we will further uncover the mystery of its molecular mechanisms, allowing this immune "signal messenger" to play a greater role in safeguarding human health.











