Decoding IL-6: From Core of Immune Regulation to Breakthroughs in Clinical Application Value
In the complex immune regulatory network of the human body, Interleukin-6 (IL-6) has always occupied a central hub position. As a multifunctional cytokine, it not only serves as a critical bridge linking innate immunity and adaptive immunity, but also plays an irreplaceable role in multiple physiological processes such as inflammatory responses, tissue repair, and hematopoietic regulation. With the rapid development of biotechnology, the mechanism of action of IL-6 has been gradually decoded, and its application value in clinical diagnosis and treatment has become increasingly prominent, making it a research hotspot and a focal point of technological translation in the field of biomedicine.
I. Brief Introduction to IL-6: A "Versatile Player" in the Immune Network
IL-6 is a cytokine first discovered by Kishimoto’s team in 1980. Initially named B-cell Stimulatory Factor 2, it earned this designation for its potent ability to promote the proliferation, differentiation and antibody production of B cells. Subsequent studies have confirmed that IL-6 is a glycoprotein with a molecular weight of approximately 26 kDa, which is mainly secreted by a variety of cells including monocytes/macrophages, T cells, B cells, fibroblasts and endothelial cells under conditions such as inflammatory stimulation, microbial infection or immune activation.
Compared with other cytokines, IL-6 is characterized by its remarkable "multifunctionality": it can not only regulate the growth, differentiation and activation of immune cells, participating in the initiation and amplification of immune responses; but also act on non-immune cells such as those in the liver, bone marrow and adipose tissue, regulating the synthesis of acute-phase response proteins, the proliferation of hematopoietic stem cells and metabolic homeostasis. This broad range of actions makes IL-6 a crucial "signal hub" connecting immunity, inflammation and metabolism, and the abnormal expression level of IL-6 is closely associated with the occurrence and development of various diseases.
II. Modes of Action and Characteristics of IL-6: Dual Attributes of Precise Regulation and Cascade Amplification
The modes of action of IL-6 are characterized by distinct specificity and high efficiency, exerting its functions mainly through three pathways: paracrine, autocrine and endocrine. The paracrine pathway mainly acts on adjacent cells in the local tissue microenvironment, participating in local inflammatory responses and tissue repair. The autocrine pathway refers to IL-6 secreted by a cell acting on the cell itself, forming a self-activating positive feedback loop. The endocrine pathway involves IL-6 entering the blood circulation to act on distal organs (e.g., liver and bone marrow), regulating physiological processes at the systemic level.
Its core characteristics are reflected in the dual attributes of "precise recognition" and "cascade amplification": On the one hand, IL-6 must bind to the specific receptor (IL-6R) on the surface of target cells to initiate downstream signal transduction. Since IL-6R is only expressed on the surface of specific cells such as hepatocytes and immune cells, the targeting of its action is ensured. On the other hand, after IL-6 binds to IL-6R, it further recruits gp130 molecules to form a heterodimer complex, triggering the activation of downstream signaling pathways. Through cascade reactions, efficient signal amplification is achieved, and a small amount of IL-6 can induce significant biological effects. In addition, the action of IL-6 is bidirectional: at physiological concentrations, it participates in the body’s normal immune regulation and tissue repair; in pathological states, however, overexpressed IL-6 can induce chronic inflammation and immune disorders, driving disease progression.
III. IL-6-Mediated Signaling Pathways and Mechanisms: A Regulatory Network Driven by Core Pathways
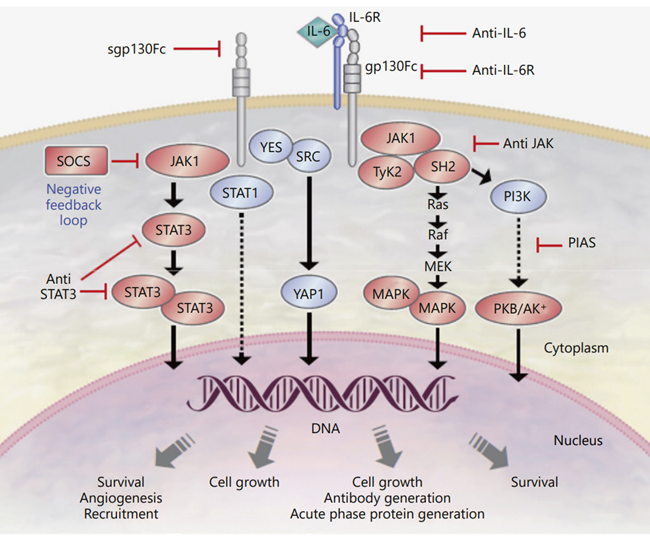
The biological functions of IL-6 are primarily mediated through three core signaling pathways, namely the JAK-STAT pathway, the Ras-MAPK pathway, and the PI3K-Akt pathway. Among these, the JAK-STAT pathway serves as its predominant signal transduction route.
1.1 JAK-STAT Pathway
Upon binding of IL-6 to IL-6R on the surface of target cells, it induces the oligomerization of IL-6R and gp130, thereby activating Janus kinases (JAKs) that are associated with the intracellular domain of gp130. Activated JAKs phosphorylate the intracellular tyrosine residues of gp130, generating binding sites for STAT proteins. STAT proteins (primarily STAT3) bind to phosphorylated gp130 and are subsequently phosphorylated and activated by JAKs. The activated STAT proteins form homodimers, translocate into the nucleus, and bind to the promoter regions of downstream target genes (e.g., acute-phase response protein genes, anti-apoptotic genes). This process regulates gene transcription, thereby mediating the activation of immune cells, inflammatory responses, as well as cell proliferation and survival.
1.2 Ras-MAPK Pathway
After IL-6 binds to its receptor and activates JAKs, it can also trigger the Ras-MAPK signaling pathway via the phosphorylation of Shc proteins. Phosphorylated Shc proteins form a complex with Grb2 and SOS, which in turn activates Ras proteins. Activated Ras further stimulates MAPK kinases (MEKs), and MEKs then phosphorylate and activate mitogen-activated protein kinases (MAPKs, such as ERK1/2). Phosphorylated MAPKs translocate into the nucleus to regulate the expression of transcription factors including c-Fos and c-Jun, participating in the modulation of cell proliferation and differentiation. This pathway plays a particularly critical role in the activation and maturation of T cells and B cells.
1.3 PI3K-Akt Pathway
JAK activation can also initiate the PI3K-Akt pathway. Activated JAKs phosphorylate insulin receptor substrates (IRSs), promoting their binding to and activation of PI3K. PI3K catalyzes the conversion of phosphatidylinositol-4,5-bisphosphate (PIP2) to phosphatidylinositol-3,4,5-trisphosphate (PIP3). PIP3 recruits Akt to the cell membrane and enables its phosphorylation and activation by PDK1. Activated Akt regulates cell survival, metabolism, and proliferation through phosphorylating downstream target proteins (e.g., Bad, mTOR). Meanwhile, it can enhance the activation of STAT3, thereby further amplifying the signaling effects of IL-6.
IV. Clinical Applications of IL-6: Full-Chain Value from Diagnostic Biomarker to Targeted Therapy
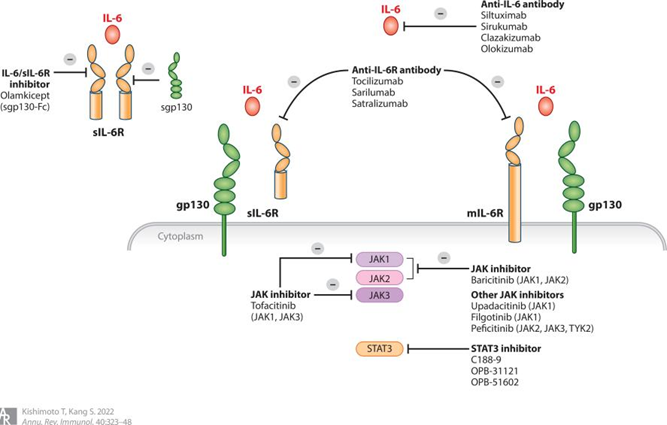
Based on the core role of IL-6 in the pathogenesis and progression of diseases, its clinical applications have expanded across multiple fields including disease diagnosis, disease monitoring, and targeted therapy, emerging as a key translational direction in the field of biomedicine.
4.1 Biomarker for Disease Diagnosis and Monitoring
As a vital inflammatory biomarker, alterations in the serum level of IL-6 are closely correlated with the onset and progression of various diseases, rendering it a crucial indicator for disease diagnosis, severity assessment, and prognostic evaluation. In sepsis, IL-6 serves as a key biomarker for early diagnosis: serum IL-6 levels in sepsis patients rise rapidly after the onset of illness, and the magnitude of elevation is positively correlated with disease severity. Compared with traditional markers such as C-reactive protein (CRP) and procalcitonin (PCT), IL-6 exhibits changes at an earlier stage, providing a basis for the early intervention of sepsis. In addition, sustained elevation of IL-6 levels is observed in autoimmune diseases including rheumatoid arthritis and systemic lupus erythematosus, which can be used to monitor disease activity. In the field of oncology, high IL-6 expression is associated with the progression, metastasis, and poor prognosis of tumors such as lung cancer, breast cancer, and multiple myeloma, serving as an auxiliary indicator for tumor prognostic assessment.
4.2 Therapeutic Agents Targeting IL-6
Targeted therapy against IL-6 and its receptor has been a research hotspot in recent years, with multiple drugs approved for clinical use. The most representative ones are anti-IL-6R monoclonal antibodies (e.g., tocilizumab) and anti-IL-6 monoclonal antibodies (e.g., siltuximab). Tocilizumab specifically binds to IL-6R, preventing the binding of IL-6 to its receptor and thereby blocking the activation of downstream signaling pathways. It has been approved for the treatment of rheumatoid arthritis, systemic juvenile idiopathic arthritis, and sepsis, significantly improving inflammatory symptoms and prognosis in patients. Siltuximab directly binds to IL-6 and inhibits its biological activity, and is approved for the treatment of Castleman’s disease and other disorders associated with excessive IL-6 expression. In addition, small-molecule inhibitors targeting the JAK-STAT pathway (e.g., tofacitinib) can also inhibit IL-6 signal transduction and are used for the treatment of autoimmune diseases, providing more options for clinical therapy.
4.3 Exploration of Other Clinical Applications
Beyond diagnosis and treatment, IL-6 has shown potential application value in regenerative medicine, vaccine development, and other fields. In tissue repair, IL-6 can promote fibroblast proliferation and collagen synthesis, accelerating wound healing, and related research has entered the preclinical stage. In vaccine development, IL-6 can be used as an immune adjuvant to enhance vaccine-induced immune responses and improve the protective efficacy of vaccines, and relevant explorations are currently underway in the development of various vaccines.
Conclusion
As a core molecule in the immune regulatory network, the mechanism of action and clinical value of IL-6 are under continuous in-depth exploration. From a precise biomarker for disease diagnosis to a key target for targeted therapy, IL-6 has become an important bridge connecting basic research and clinical applications. With the continuous advancement of biotechnology, it is believed that future technological translations centered on IL-6 will achieve continuous breakthroughs, bringing new therapeutic strategies for various diseases such as inflammatory diseases, autoimmune diseases, and tumors, and safeguarding human health.
I. Brief Introduction to IL-6: A "Versatile Player" in the Immune Network
IL-6 is a cytokine first discovered by Kishimoto’s team in 1980. Initially named B-cell Stimulatory Factor 2, it earned this designation for its potent ability to promote the proliferation, differentiation and antibody production of B cells. Subsequent studies have confirmed that IL-6 is a glycoprotein with a molecular weight of approximately 26 kDa, which is mainly secreted by a variety of cells including monocytes/macrophages, T cells, B cells, fibroblasts and endothelial cells under conditions such as inflammatory stimulation, microbial infection or immune activation.
Compared with other cytokines, IL-6 is characterized by its remarkable "multifunctionality": it can not only regulate the growth, differentiation and activation of immune cells, participating in the initiation and amplification of immune responses; but also act on non-immune cells such as those in the liver, bone marrow and adipose tissue, regulating the synthesis of acute-phase response proteins, the proliferation of hematopoietic stem cells and metabolic homeostasis. This broad range of actions makes IL-6 a crucial "signal hub" connecting immunity, inflammation and metabolism, and the abnormal expression level of IL-6 is closely associated with the occurrence and development of various diseases.
II. Modes of Action and Characteristics of IL-6: Dual Attributes of Precise Regulation and Cascade Amplification
The modes of action of IL-6 are characterized by distinct specificity and high efficiency, exerting its functions mainly through three pathways: paracrine, autocrine and endocrine. The paracrine pathway mainly acts on adjacent cells in the local tissue microenvironment, participating in local inflammatory responses and tissue repair. The autocrine pathway refers to IL-6 secreted by a cell acting on the cell itself, forming a self-activating positive feedback loop. The endocrine pathway involves IL-6 entering the blood circulation to act on distal organs (e.g., liver and bone marrow), regulating physiological processes at the systemic level.
Its core characteristics are reflected in the dual attributes of "precise recognition" and "cascade amplification": On the one hand, IL-6 must bind to the specific receptor (IL-6R) on the surface of target cells to initiate downstream signal transduction. Since IL-6R is only expressed on the surface of specific cells such as hepatocytes and immune cells, the targeting of its action is ensured. On the other hand, after IL-6 binds to IL-6R, it further recruits gp130 molecules to form a heterodimer complex, triggering the activation of downstream signaling pathways. Through cascade reactions, efficient signal amplification is achieved, and a small amount of IL-6 can induce significant biological effects. In addition, the action of IL-6 is bidirectional: at physiological concentrations, it participates in the body’s normal immune regulation and tissue repair; in pathological states, however, overexpressed IL-6 can induce chronic inflammation and immune disorders, driving disease progression.
III. IL-6-Mediated Signaling Pathways and Mechanisms: A Regulatory Network Driven by Core Pathways

The biological functions of IL-6 are primarily mediated through three core signaling pathways, namely the JAK-STAT pathway, the Ras-MAPK pathway, and the PI3K-Akt pathway. Among these, the JAK-STAT pathway serves as its predominant signal transduction route.
1.1 JAK-STAT Pathway
Upon binding of IL-6 to IL-6R on the surface of target cells, it induces the oligomerization of IL-6R and gp130, thereby activating Janus kinases (JAKs) that are associated with the intracellular domain of gp130. Activated JAKs phosphorylate the intracellular tyrosine residues of gp130, generating binding sites for STAT proteins. STAT proteins (primarily STAT3) bind to phosphorylated gp130 and are subsequently phosphorylated and activated by JAKs. The activated STAT proteins form homodimers, translocate into the nucleus, and bind to the promoter regions of downstream target genes (e.g., acute-phase response protein genes, anti-apoptotic genes). This process regulates gene transcription, thereby mediating the activation of immune cells, inflammatory responses, as well as cell proliferation and survival.
1.2 Ras-MAPK Pathway
After IL-6 binds to its receptor and activates JAKs, it can also trigger the Ras-MAPK signaling pathway via the phosphorylation of Shc proteins. Phosphorylated Shc proteins form a complex with Grb2 and SOS, which in turn activates Ras proteins. Activated Ras further stimulates MAPK kinases (MEKs), and MEKs then phosphorylate and activate mitogen-activated protein kinases (MAPKs, such as ERK1/2). Phosphorylated MAPKs translocate into the nucleus to regulate the expression of transcription factors including c-Fos and c-Jun, participating in the modulation of cell proliferation and differentiation. This pathway plays a particularly critical role in the activation and maturation of T cells and B cells.
1.3 PI3K-Akt Pathway
JAK activation can also initiate the PI3K-Akt pathway. Activated JAKs phosphorylate insulin receptor substrates (IRSs), promoting their binding to and activation of PI3K. PI3K catalyzes the conversion of phosphatidylinositol-4,5-bisphosphate (PIP2) to phosphatidylinositol-3,4,5-trisphosphate (PIP3). PIP3 recruits Akt to the cell membrane and enables its phosphorylation and activation by PDK1. Activated Akt regulates cell survival, metabolism, and proliferation through phosphorylating downstream target proteins (e.g., Bad, mTOR). Meanwhile, it can enhance the activation of STAT3, thereby further amplifying the signaling effects of IL-6.
IV. Clinical Applications of IL-6: Full-Chain Value from Diagnostic Biomarker to Targeted Therapy

Based on the core role of IL-6 in the pathogenesis and progression of diseases, its clinical applications have expanded across multiple fields including disease diagnosis, disease monitoring, and targeted therapy, emerging as a key translational direction in the field of biomedicine.
4.1 Biomarker for Disease Diagnosis and Monitoring
As a vital inflammatory biomarker, alterations in the serum level of IL-6 are closely correlated with the onset and progression of various diseases, rendering it a crucial indicator for disease diagnosis, severity assessment, and prognostic evaluation. In sepsis, IL-6 serves as a key biomarker for early diagnosis: serum IL-6 levels in sepsis patients rise rapidly after the onset of illness, and the magnitude of elevation is positively correlated with disease severity. Compared with traditional markers such as C-reactive protein (CRP) and procalcitonin (PCT), IL-6 exhibits changes at an earlier stage, providing a basis for the early intervention of sepsis. In addition, sustained elevation of IL-6 levels is observed in autoimmune diseases including rheumatoid arthritis and systemic lupus erythematosus, which can be used to monitor disease activity. In the field of oncology, high IL-6 expression is associated with the progression, metastasis, and poor prognosis of tumors such as lung cancer, breast cancer, and multiple myeloma, serving as an auxiliary indicator for tumor prognostic assessment.
4.2 Therapeutic Agents Targeting IL-6
Targeted therapy against IL-6 and its receptor has been a research hotspot in recent years, with multiple drugs approved for clinical use. The most representative ones are anti-IL-6R monoclonal antibodies (e.g., tocilizumab) and anti-IL-6 monoclonal antibodies (e.g., siltuximab). Tocilizumab specifically binds to IL-6R, preventing the binding of IL-6 to its receptor and thereby blocking the activation of downstream signaling pathways. It has been approved for the treatment of rheumatoid arthritis, systemic juvenile idiopathic arthritis, and sepsis, significantly improving inflammatory symptoms and prognosis in patients. Siltuximab directly binds to IL-6 and inhibits its biological activity, and is approved for the treatment of Castleman’s disease and other disorders associated with excessive IL-6 expression. In addition, small-molecule inhibitors targeting the JAK-STAT pathway (e.g., tofacitinib) can also inhibit IL-6 signal transduction and are used for the treatment of autoimmune diseases, providing more options for clinical therapy.
4.3 Exploration of Other Clinical Applications
Beyond diagnosis and treatment, IL-6 has shown potential application value in regenerative medicine, vaccine development, and other fields. In tissue repair, IL-6 can promote fibroblast proliferation and collagen synthesis, accelerating wound healing, and related research has entered the preclinical stage. In vaccine development, IL-6 can be used as an immune adjuvant to enhance vaccine-induced immune responses and improve the protective efficacy of vaccines, and relevant explorations are currently underway in the development of various vaccines.
Conclusion
As a core molecule in the immune regulatory network, the mechanism of action and clinical value of IL-6 are under continuous in-depth exploration. From a precise biomarker for disease diagnosis to a key target for targeted therapy, IL-6 has become an important bridge connecting basic research and clinical applications. With the continuous advancement of biotechnology, it is believed that future technological translations centered on IL-6 will achieve continuous breakthroughs, bringing new therapeutic strategies for various diseases such as inflammatory diseases, autoimmune diseases, and tumors, and safeguarding human health.











